Mesenchymal stromal cells, also known as MSCs, are versatile cell populations derived from fetal and adult tissues. MSCs possess differentiation potential into various skeletal lineages such as osteogenic, chondrogenic, and tenogenic lineages. Furthermore, they originate from embryonic mesoderm, similar to limb bud mesodermal cells.
While MSCs can differentiate into skeletal-related lineages in vitro, their ability to self-organize into complex skeletal structures or connective tissues, as in the limb, has not been demonstrated. In a recent study published in the World Journal of Stem Cells, researchers investigated the expression of molecular markers associated with MSC commitment to skeletal lineages and their potential to generate skeletal elements in vivo.
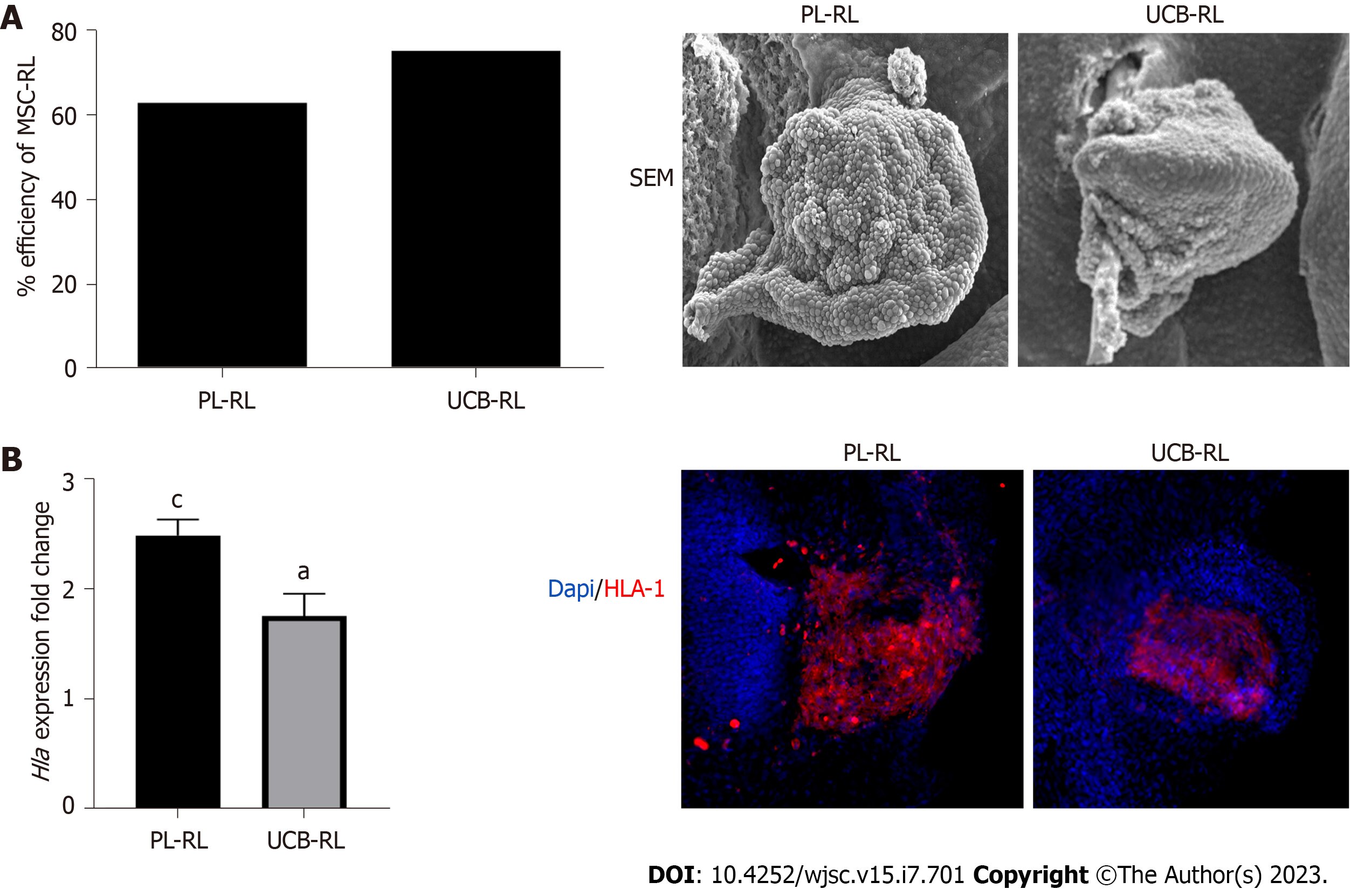
The study utilized the recombinant limb (RL) system to evaluate the differentiation and patterning potential of MSCs. The researchers used dissociated-reaggregated MSCs from human placenta (PL) and umbilical cord blood (UCB), which were then introduced into an ectodermal cover derived from a chicken limb bud. The filled ectoderm with the reaggregated cells was subsequently grafted into a chick embryo.
Under these experimental conditions, MSCs received signals from the ectoderm and responded by differentiating and patterning into skeletal elements. Various techniques such as quantitative polymerase chain reaction, histology, immunofluorescence, scanning electron microscopy, and in situ hybridization were employed to evaluate the differentiation and morphogenetic signals of the MSCs.
The research findings revealed that both PL-MSCs and UCB-MSCs constituting the RLs expressed molecular markers associated with chondrogenic, osteogenic, and tenogenic lineages. However, despite their commitment to differentiate into these lineages, the MSCs could not generate complex skeletal structures in vivo. Additionally, the UCB-MSCs showed a preference for osteogenic commitment and a tenogenic cell fate.
These results suggest that the commitment of MSCs to differentiate into skeletal lineages depends on the cell source and is not sufficient to generate complex skeletal structures in vivo. The findings also indicate that the intrinsic characteristics of MSCs may contribute to the failure to form skeletal elements. Thus, further research is necessary to fully understand the biological aspects of MSCs and their response to morphogenetic signals in an in vivo context.
In conclusion, the study demonstrates that although PL-MSCs and UCB-MSCs express molecular markers associated with differentiation into skeletal lineages, they lack the capacity to generate complex skeletal structures in vivo.
More information:
Jessica Cristina Marín-Llera et al, Commitment of human mesenchymal stromal cells to skeletal lineages is independent of their morphogenetic capacity, World Journal of Stem Cells (2023). DOI: 10.4252/wjsc.v15.i7.701
Provided by
World Journal of Stem Cells
Citation:
Commitment of human mesenchymal stromal cells to skeletal lineages is independent of their morphogenetic capacity: Study (2023, July 26)
retrieved 26 July 2023
from https://phys.org/news/2023-07-commitment-human-mesenchymal
Denial of responsibility! TechCodex is an automatic aggregator of the all world’s media. In each content, the hyperlink to the primary source is specified. All trademarks belong to their rightful owners, and all materials to their authors. For any complaint, please reach us at – [email protected]. We will take necessary action within 24 hours.

Jessica Irvine is a tech enthusiast specializing in gadgets. From smart home devices to cutting-edge electronics, Jessica explores the world of consumer tech, offering readers comprehensive reviews, hands-on experiences, and expert insights into the coolest and most innovative gadgets on the market.
Denial of responsibility! TechCodex is an automatic aggregator of Global media. In each content, the hyperlink to the primary source is specified. All trademarks belong to their rightful owners, and all materials to their authors. For any complaint, please reach us at – [email protected]. We will take necessary action within 24 hours.