Green hydrogen production through the direct splitting of water using solar energy requires highly efficient photoelectrodes that possess multiple capabilities simultaneously. These photoelectrodes should excel at converting sunlight into electricity, remain stable in acidic or basic water, act as catalysts to facilitate water splitting, and be affordable, abundant, and non-toxic. One promising material class for this purpose is metal oxides.
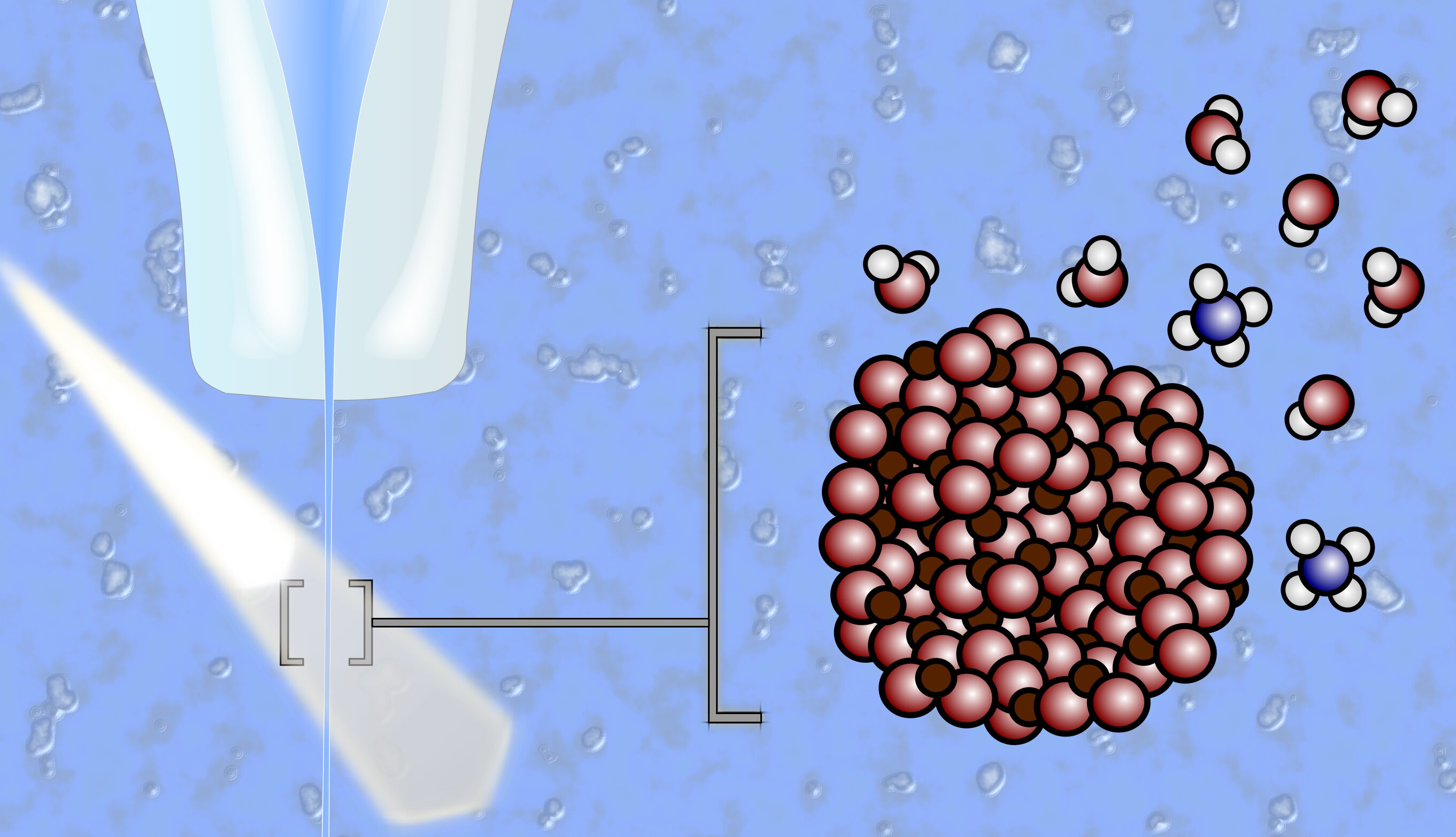
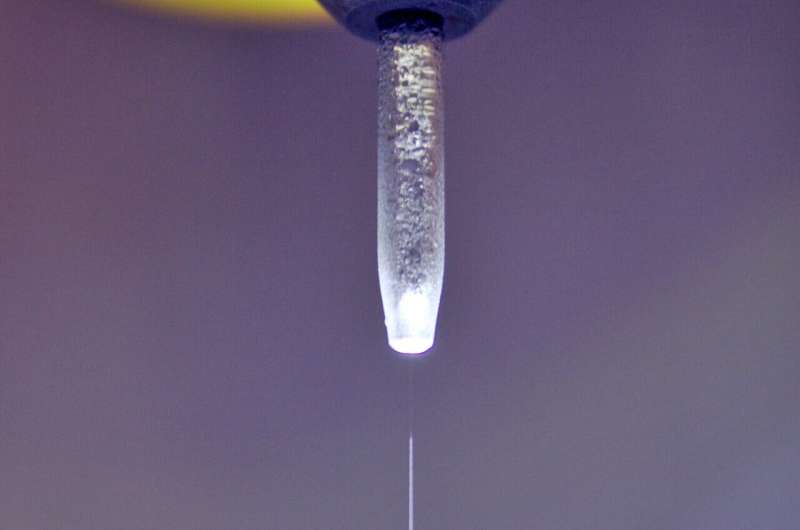
However, investigating the interactions between solid metal oxide electrodes and aqueous electrolytes presents challenges. Standard X-ray analysis methods are ineffective in liquid environments. In such cases, experiments involving a liquid jet, where metal oxide nanoparticles are suspended, prove to be suitable.
At BESSY II, researchers employ this liquid jet method to study the interfaces between metal oxide particles and liquid electrolytes. By passing the liquid jet through X-ray light, the interference of evaporated molecules with the measurement data is minimized. Dr. Robert Seidel, an expert in this method, has used it to conduct experiments at BESSY II alongside Dr. Hebatallah Ali and Dr. Bernd Winter from the Fritz Haber Institute. They focus on investigating two important model systems involving iron oxide (hematite, α-Fe2O3) and anatase (titanium oxide or TiO2) nanoparticles in aqueous electrolytes with varying pH values. These model systems are photocatalytic and enable the study of solid/electrolyte interfaces at the molecular level, as well as the exploration of chemical reactions at electrode-electrolyte interfaces.
The researchers employ resonant photoelectron spectroscopy (PES) to identify unique reaction fingerprints, enabling them to reconstruct the formation of reaction products under different conditions, particularly as affected by pH. The researchers find that the pH value of the electrolyte significantly impacts the reactions occurring on the surfaces of hematite and anatase nanoparticles. At low pH, water molecules on the surface of hematite tend to split, while anatase nanoparticles adsorb water molecules without splitting. Water molecules require a basic pH value to break down on anatase nanoparticles. These insights into surface-water interactions are only possible through the liquid-jet method.
The spectroscopic analysis also reveals ultra-fast electron transitions between metal oxide nanoparticles and water molecules on their surfaces. These findings provide valuable insights into the initial stages of water dissociation and contribute to understanding the mechanisms of light-induced water splitting on metal oxide surfaces.
The special issue ‘Applications of Liquid Microjets in Chemistry’:
To study water surfaces without disturbances caused by interloping molecules, researchers have long faced challenges. In 1988, the liquid jet method was introduced, enabling the study of water surfaces without such interference. The microjet is a fast-flowing stream of liquid that is so narrow it produces an extremely dilute vapor cloud. This allows photons and particles to reach and leave the surface of the jet without colliding with vapor molecules.
The above-mentioned information is drawn from a special issue of Accounts of Chemical Research, which explores the applications of liquid microjets in chemistry. The issue’s guest-editor, Dr. Robert Seidel, discusses the liquid jet method and presents new experiments conducted at BESSY II.
More information:
Hebatallah Ali et al, The Metal-Oxide Nanoparticle–Aqueous Solution Interface Studied by Liquid-Microjet Photoemission, Accounts of Chemical Research (2023). DOI: 10.1021/acs.accounts.2c00789
Citation:
BESSY II: Surface analysis of catalyst particles in aqueous solutions (2023, July 21)
retrieved 22 July 2023
from https://phys.org/news/2023-07-bessy-ii-surface-analysis-catalyst.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.